 Site-specific introduction of distinct molecules into proteins and peptides is a challenging task. It becomes even more challenging if the labeling reaction has to be performed in living systems. How can we overcome undesired cross reactions with functional groups on proteins other than our target and decrease the immunogenicity of the external starting materials? Bioorthogonal chemistry provides a solution for this kind of questions. In this commentary, I will focus on two new methods, irreversible cyclopropenone (Row. et al., 2017) and reversible RevAmine conjugation (Cowell et al. 2017) and discuss their value for bioorthogonal labeling of peptides and proteins.
Site-specific introduction of distinct molecules into proteins and peptides is a challenging task. It becomes even more challenging if the labeling reaction has to be performed in living systems. How can we overcome undesired cross reactions with functional groups on proteins other than our target and decrease the immunogenicity of the external starting materials? Bioorthogonal chemistry provides a solution for this kind of questions. In this commentary, I will focus on two new methods, irreversible cyclopropenone (Row. et al., 2017) and reversible RevAmine conjugation (Cowell et al. 2017) and discuss their value for bioorthogonal labeling of peptides and proteins.
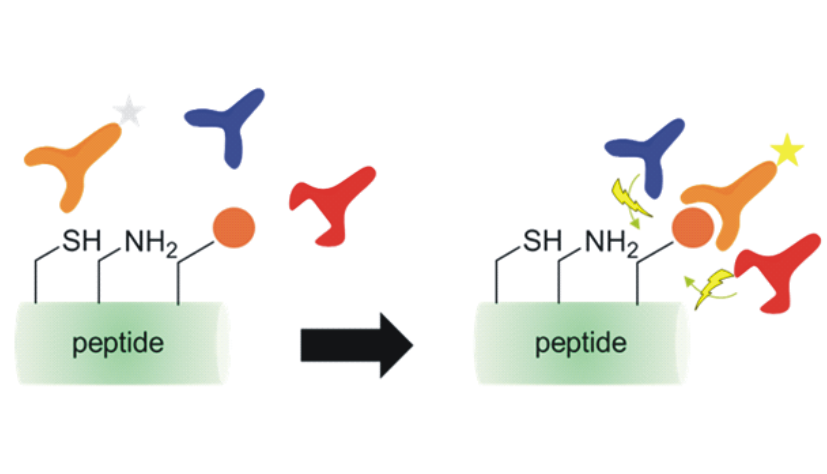
For more please download PDF file.
Contributed by Can Araman
Can Araman completed his PhD degree under the supervision of Prof. Christian Becker at the University of Vienna. During this period, he worked on the semisynthesis of prion protein variants carrying glycan mimics and investigated their biophysical/biochemical properties in vitro. Currently, he is working as a postdoctoral researcher at the Leiden Institute of Chemistry (Netherlands), Bioorganic Synthesis Division under the guidance of Dr. Sander van Kasteren. His research interests are directed towards to the study kinetics of antigen processing & presentation using bioorthogonal chemistry as well as semisynthesis of posttranslationally modified antigens.
