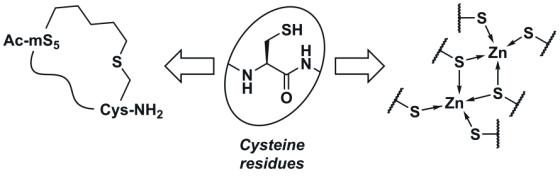
 Cysteine is not, of course, one of the ten “essential” proteinogenic α-amino acids that mammals require in their diet. In many other respects, however, cysteine is just as important as any of the “essential” amino acids (or, for that matter, any of the other nine “inessential” ones). In fact, one could go as far as to call cysteine a special amino acid because of the rich chemistry of its thiol group. It is from this richness that diverse functions of cysteine residues in proteins and peptides arise: cysteine residues form disulfide bridges; they co-ordinate metal ions; they are responsible for the hydrolytic activity of many proteases; they are sites of post-translational modification; and they are responsible for cellular redox homeostasis. In recent years, the special properties of cysteine’s thiol group have also become indispensable to synthetic chemists and chemical biologists. This short article highlights two recent publications dealing with different aspects of cysteine’s thiol chemistry. The first publication (Zhao et al., J. Peptide. Sci., 2016, 22, 540-544) sees the thiol group forming thioether cross-links in the context of a new synthetic methodology, whilst the second (Tarasava et al., Biopolymers Peptide Sci., 2016, 106, 295-308) explores the interplay of thiolate-metal bonding and disulfide bridges in the structure of a model metalloprotein. Together, these reports illustrate the importance of cysteine and its thiol group in different areas of chemistry and biology.
Cysteine is not, of course, one of the ten “essential” proteinogenic α-amino acids that mammals require in their diet. In many other respects, however, cysteine is just as important as any of the “essential” amino acids (or, for that matter, any of the other nine “inessential” ones). In fact, one could go as far as to call cysteine a special amino acid because of the rich chemistry of its thiol group. It is from this richness that diverse functions of cysteine residues in proteins and peptides arise: cysteine residues form disulfide bridges; they co-ordinate metal ions; they are responsible for the hydrolytic activity of many proteases; they are sites of post-translational modification; and they are responsible for cellular redox homeostasis. In recent years, the special properties of cysteine’s thiol group have also become indispensable to synthetic chemists and chemical biologists. This short article highlights two recent publications dealing with different aspects of cysteine’s thiol chemistry. The first publication (Zhao et al., J. Peptide. Sci., 2016, 22, 540-544) sees the thiol group forming thioether cross-links in the context of a new synthetic methodology, whilst the second (Tarasava et al., Biopolymers Peptide Sci., 2016, 106, 295-308) explores the interplay of thiolate-metal bonding and disulfide bridges in the structure of a model metalloprotein. Together, these reports illustrate the importance of cysteine and its thiol group in different areas of chemistry and biology.
For more please download PDF file.
Contributed by George Preston
George is a Research Associate at King’s College London. In his research, he is interested in using peptides to understand more about protein covalent modifications, and he currently works on the “EXPOsOMICS” project (http://www.exposomicsproject.eu/).
