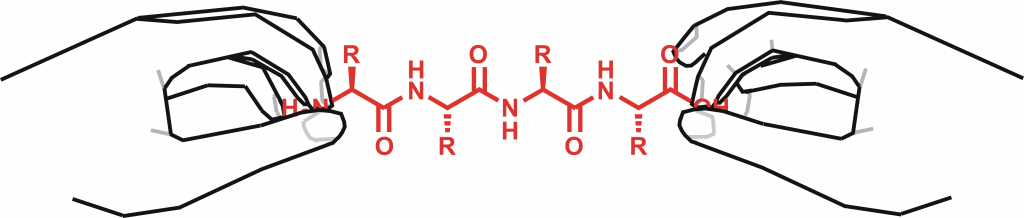
 Peptides can sometimes exhibit unusual stereochemistry. Occasionally, for example, the usual l-amino acid building block might be substituted with its mirror-image counterpart. Such features can impact on function, but may be subtle enough to evade detection via routine methods. In this article, I highlight some innovative mass-spectrometry-based approaches to the characterisation of peptides with exotic stereochemical features.
Peptides can sometimes exhibit unusual stereochemistry. Occasionally, for example, the usual l-amino acid building block might be substituted with its mirror-image counterpart. Such features can impact on function, but may be subtle enough to evade detection via routine methods. In this article, I highlight some innovative mass-spectrometry-based approaches to the characterisation of peptides with exotic stereochemical features.
For more please download PDF file.
Contributed by George Preston
George is a Research Associate at King’s College London. In his research, he is interested in using peptides to understand more about protein covalent modifications, and he currently works on the “EXPOsOMICS” project (http://www.exposomicsproject.eu/).
