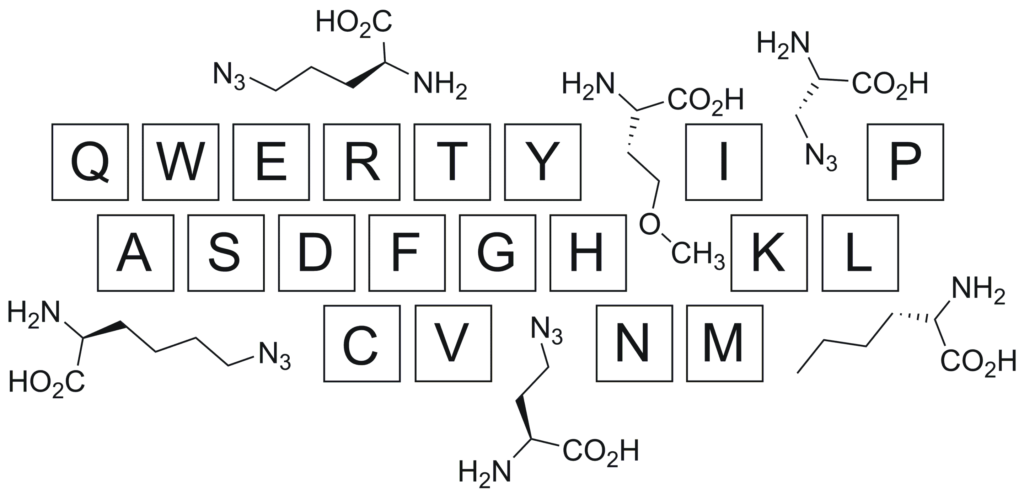
The use of non-canonical amino-acid building blocks for peptide synthesis is a flexible way of introducing functionalities not normally found in biological systems. Two recent papers on this theme, both from Journal of Peptide Science, are discussed. In the first (Grob et al., 2017), methionine analogues are used to suppress side reactions in the synthesis of a radiopharmaceutical; in the second (Pícha et al., 2017) practical considerations for syntheses of azide-containing building blocks are explored.
The use of non-canonical amino-acid building blocks for peptide synthesis is a flexible way of introducing functionalities not normally found in biological systems. Two recent papers on this theme, both from Journal of Peptide Science, are discussed. In the first (Grob et al., 2017), methionine analogues are used to suppress side reactions in the synthesis of a radiopharmaceutical; in the second (Pícha et al., 2017) practical considerations for syntheses of azide-containing building blocks are explored.
For more please download PDF file.
Contributed by George Preston
George is a Research Associate at King’s College London. In his research, he is interested in using peptides to understand more about protein covalent modifications, and he currently works on the “EXPOsOMICS” project (http://www.exposomicsproject.eu/).