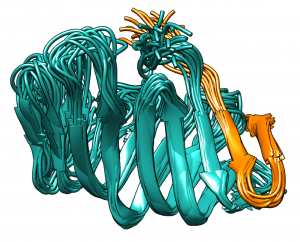
Swiss researchers have recently revealed the mechanism by which the insect-derived antimicrobial peptide thanatin targets the intermembrane protein complex required for lipopolysaccharide (LPS) transport in Escherichia coli. Employing a series of elegant techniques directed at increasing levels of magnification and ultimately, structural determination by NMR, these researchers show how thanatin interferes with assembly of the seven-member macromolecular protein complex which forms a molecular bridge supporting ATP-driven transport of LPS from the inner membrane to the outer membrane (Vetterli et al, 2018).
Swiss researchers have recently revealed the mechanism by which the insect-derived antimicrobial peptide thanatin targets the intermembrane protein complex required for lipopolysaccharide (LPS) transport in Escherichia coli. Employing a series of elegant techniques directed at increasing levels of magnification and ultimately, structural determination by NMR, these researchers show how thanatin interferes with assembly of the seven-member macromolecular protein complex which forms a molecular bridge supporting ATP-driven transport of LPS from the inner membrane to the outer membrane (Vetterli et al, 2018).
For more please download PDF file.
Contributed by Susan J. Tzotzos
Susan Tzotzos works for the Vienna-based biotech company Apeptico Forschung & Entwicklung GmbH, where she designs therapeutic peptides, oversees their synthesis and follows up their application in clinical trials. Susan also manages various research collaborations with university departments carrying out projects investigating the mode of action of Apeptico’s therapeutic peptides.